|
4/1/2024 0 Comments Dalton atomic theory modelIf a substance is burnt, phlogiston evolves and goes into atmosphere and clax (meaning a non-useful thing) is left behind as an ash. The theory stated that the substance undergoing combustion mainly contains a mysterious matter called phlogiston and some clax. Previously, we defined an atom as the smallest part of an element that maintains the identity of that element. 1 ), is a fundamental concept that states that all elements are composed of atoms. changeĭuring chemical change both physical and chemical properties of the substance change.Įxamples: Freezing of water, melting of ice, Heating of tungsten wire to red hot condition.Įxamples: Rusting of iron, burning of wood, digestion of food, etc. Rutherford model, description of the structure of atoms proposed (1911) by the New Zealand-born physicist Ernest Rutherford. The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 4.2.1 4.2. There is enthalpy change in chemical change.ĭuring physical change physical properties like phase, texture, shape, size, etc. There is negligible change in enthalpy of the system in physical change. In a chemical change, energy is produced (heat, light, sound, etc.) In a physical change, no energy is produced. Physical change is reversible and original substance can be recovered.Ĭhemical change is irreversible and original substance cannot be recovered. The molecular composition of a substance changes completely There is only rearrangement of molecules while their actual composition remains same. Physical changes are those changes in which there is an only change in the appearance of the substance but no change in its chemical composition.Ĭhemical changes are those changes which cause a change in chemical composition of a substance.Įntirely new substance (one or more) is formed. Rutherford’s work helped to establish the nuclear model of the atom and paved the way for the development of nuclear weapons and energy.Distinguishing Between Physical Changes and Chemical Changes: Physical Change Rutherford performed many groundbreaking experiments on the structure of the atom, including the famous gold foil experiment in which he demonstrated that the atom has a small, dense nucleus at its center. Bohr’s model helped to explain the structure and behavior of atoms in terms of quantum mechanics and is still widely used today.Įrnest Rutherford was a New Zealand-born physicist who is considered the father of nuclear physics. Bohr developed the Bohr model of the atom, which describes the atom as a small, positively charged nucleus surrounded by orbiting electrons. Niels Bohr was a Danish physicist who made important contributions to the development of quantum mechanics and the modern atomic theory. Dalton’s atomic theory was a major breakthrough in understanding the nature of matter and laid the foundation for modern atomic theory. Dalton developed the theory in the early 1800s, based on his observations that all atoms of a given element are identical and that atoms combine in fixed ratios to form molecules. John Dalton was an English chemist and physicist who is best known for his work on the modern atomic theory. However, there are a few key figures whose work was essential in developing the theory. The modern atomic theory is based on the work of many scientists over many years. Modern science has shown that atoms can exchange electrons to form chemical bonds. Modern science has shown that the atom is made up of protons, neutrons, and electrons, which are arranged in shells and orbitals.ģ) Dalton’s atomic theory does not account for the behavior of atoms in chemical reactions. However, modern science has shown that atoms can be divided into subatomic particles (protons, neutrons, and electrons).Ģ) Dalton’s atomic theory does not account for the structure of the atom. Limitations of Dalton’s Atomic Theoryĭalton’s atomic theory has several limitations.ġ) Dalton’s atomic theory is based on the idea that atoms are indivisible and indestructible. 
Chemical reactions involve the rearrangement of atoms. Atoms of different elements are different.ĥ. Atoms of the same element are identical.Ĥ. Atoms are indivisible and indestructible.ģ. 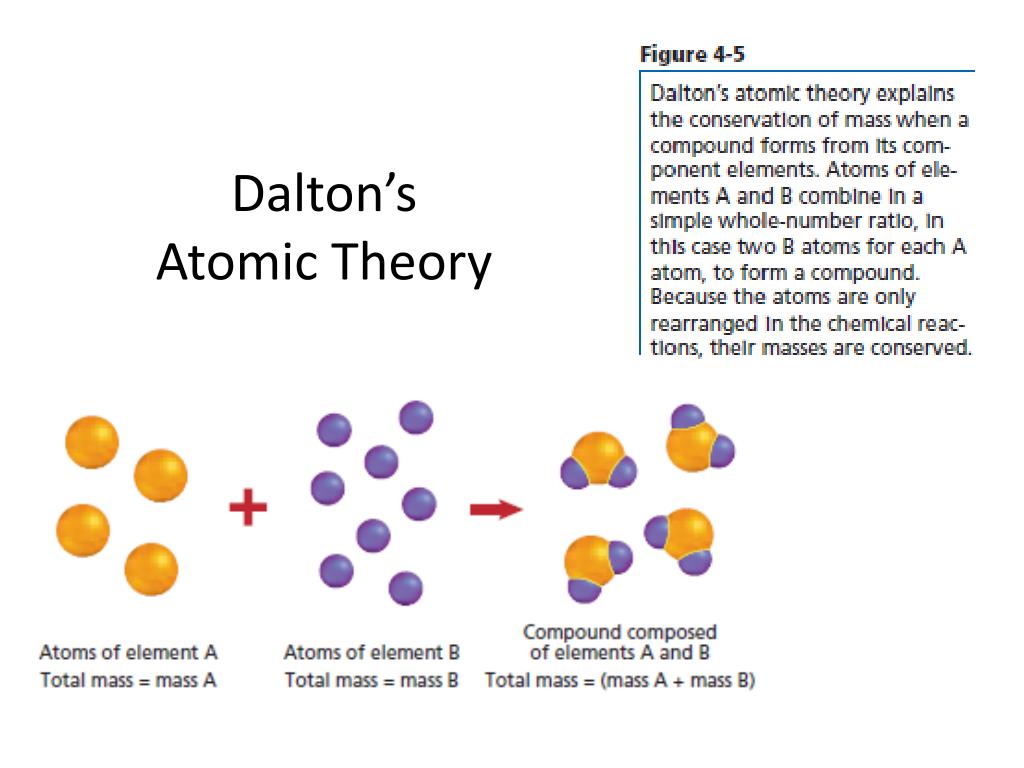
The postulates of Dalton’s atomic theory are:Ģ. Dalton also proposed that atoms of the same element are identical and that atoms combine to form molecules. Dalton proposed that atoms are indivisible and that atoms of different elements have different masses. Dalton’s atomic model is a model of the atom in which the atom is composed of small particles called atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
